I’ve been reading some excellent articles by Bruce Lamphear (https://blanphear.substack.com/) about the epidemics of chemical mayhem assaulting our communities. He has written a poem, Threshold, which is so terribly apt when talking about lead poisoning:
They tell us
toxic chemicals have a threshold—
a line
below which harm
does not begin.
But what if the line
is only hope,
drawn to steady our hands?
What if harm
does not arrive,
but seeps—
into blood,
into bone,
disrupting the quiet
work of cells?
If there is no safe edge,
no place untouched,
only degrees of injury—
how many lives
are shortened,
damaged,
or broken
before we finally say
enough?
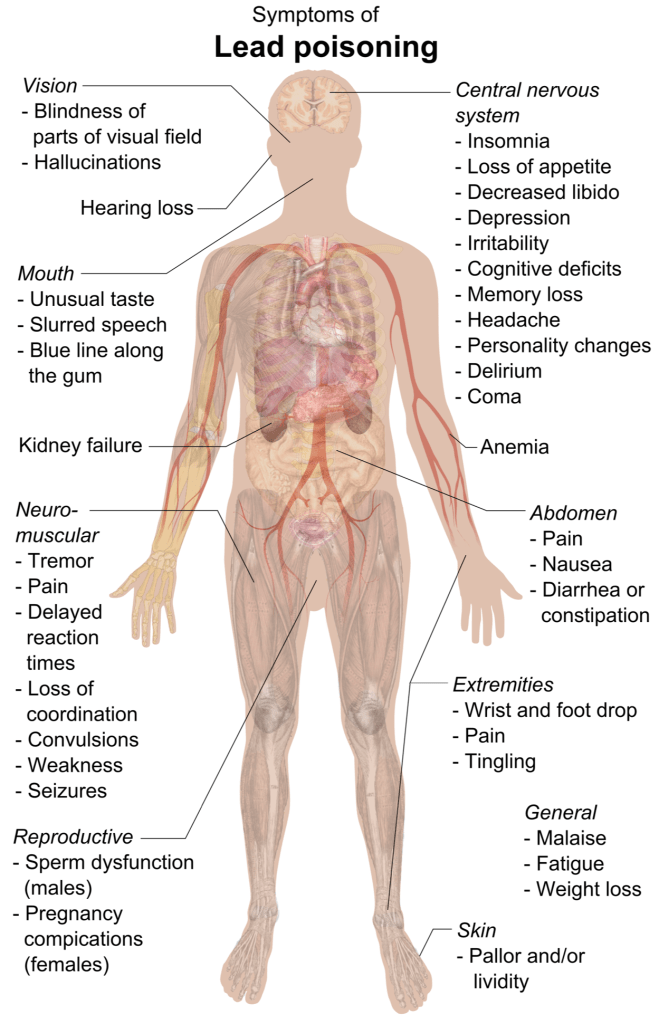
Lead poisoning is subtle and unavoidable; it demands that we sacrifice our health and our children’s futures, it has made liars and murderers out of the people that ought to be protecting us and I believe that lead poisoning is preventable.
My journey with lead started many years ago, but it was the news that my nephew was working in a lead smelter that heralded the end of my retirement and a new study of all things related to lead. I have now spent several years on an extensive literature review on lead poisoning, treatments, geological distribution of lead, interaction with health authorities and workplace health regulators, and looking at the history of lead poisoning.
Lead exposure is unavoidable, but efforts to reduce the health effects of lead have concentrated almost exclusively on reducing exposure, not prevention. We have the uncomfortable truth that reducing exposure to lead is nearly impossible because there are so many sources of lead in our environment.
Lead is a biotoxin that accumulates in our bodies, either quickly or slowly depending on exposure, and as we age, the lead-caused medical conditions increase.
As things stand, we can’t avoid the health impacts caused by lead exposure.
Lead in the body is dangerous for many reasons:
- lead mimics calcium in the body. It can take the place of calcium, so it inhibits calcium channel operations, causing neurological dysfunction, and is incorporated into bone, weakening bone and becoming a toxic store of lead in the body that will be released by inflammation, cancer, osteoporosis, pregnancy and lactation.
- lead can displace other metals such as zinc which causes inhibition of zinc enzymes.
- lead binds to sulphur, inhibiting sulphur-enzymes and -anti-oxidants.
- lead is a strong inflammatory agent, causing inflammation and calcification in blood vessels and other organs such as the brain and prostate.
- lead is a potent oxidizing agent that damages cell membranes and DNA and can cause cancer.
Given this list it is clear that “you do not want lead in your body because you will be harmed“. There is no safe threshold.
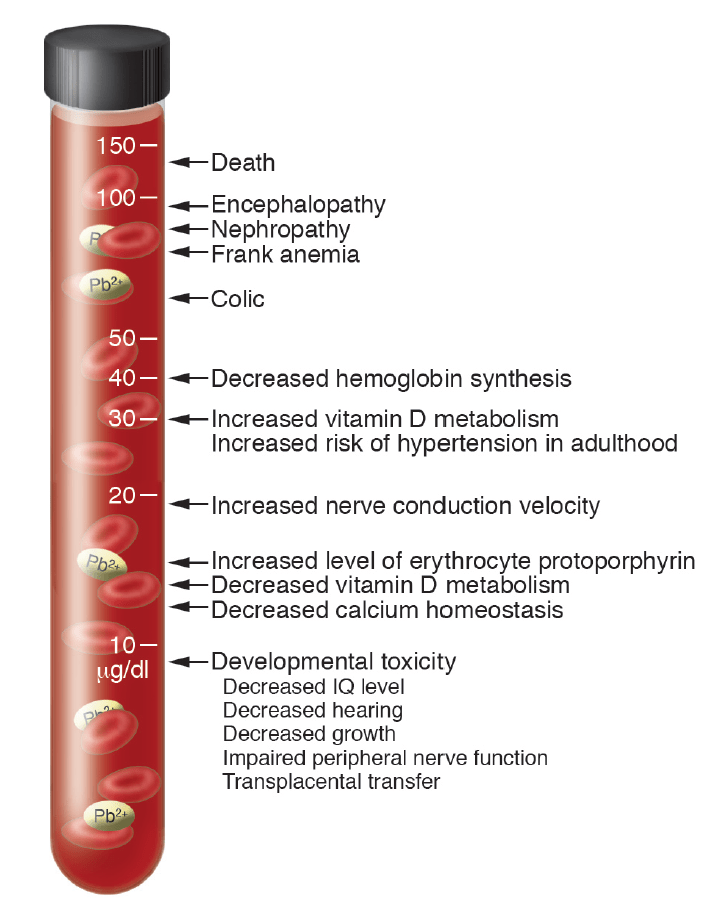
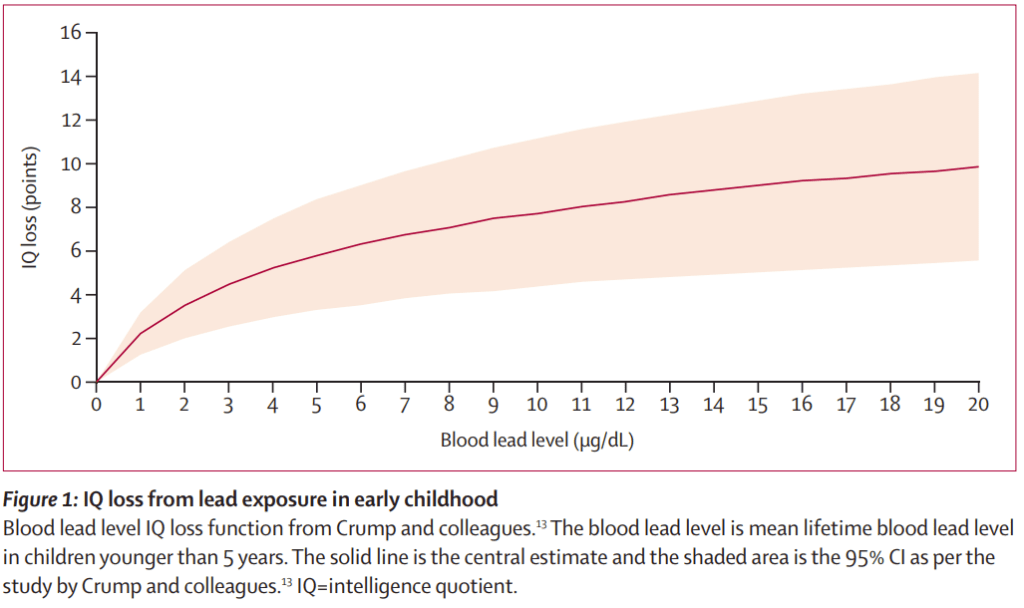
The notification threshold of 5ug/dL is simply a number a committee chose that is acceptable to people who want it to be higher and people who know enough to want it lower. Neurological effects begin to be apparent at levels as low as 2 ug/dL which is a reminder that 5ug/dL is most certainly not safe.
If you’re a lead risk worker, then the industry has demanded that the acceptable level of blood lead is 20 ug/dL and action isn’t required until it exceeds 30ug/dL, which is without question harmful in the short and long term. Lead risk workers are being paid to sacrifice their health, and in many cases, to die early.
But tolerating or ignoring the effects of lead is a legacy reaction. It isn’t necessary and the effects of exposure to lead can be avoided/
There are chemical compounds, chelators, that remove lead from the body, preventing the accumulation of lead and in essence providing immunity to the effects of lead.
We’ve been able to remove lead with chelators for the last 70 years but for one reason or the other, manipulation, disinformation or ignorance, it simply hasn’t happened except on a very small scale.
It is almost unbelievable that one of the most promising solutions for preventing harm from lead poisoning has been effectively blacklisted by most of the medical community. The worst part of this is that the reasons given are either dated or unfounded, often due to ignorance. Urban legends exist in Medicine, and they are almost always harmful.
I’d been in contact with a father in Austria who was concerned when he discovered he and his family had been exposed to lead paint while he was renovating their 1950s home. One of the doctors he approached made this comment when chelation was mentioned: that’s nasty stuff.
That’s pretty close-minded and dismissive, but many physicians are simply not aware that the risk from lead in lead paint is still there in millions of old houses in Australia, that the risk from lead from cars burning leaded petro/gasoline is still present in the attics and gardens of buildings lining our roadways, that the risk of lead from mining/smelting is still there, as well as the risk of lead in spices and folk medicines. The least scathing comment I can make about our medical system is that their complacency has led them to believe lead is no longer a problem.
I’m going to try and give you a feel for what this complacency means.
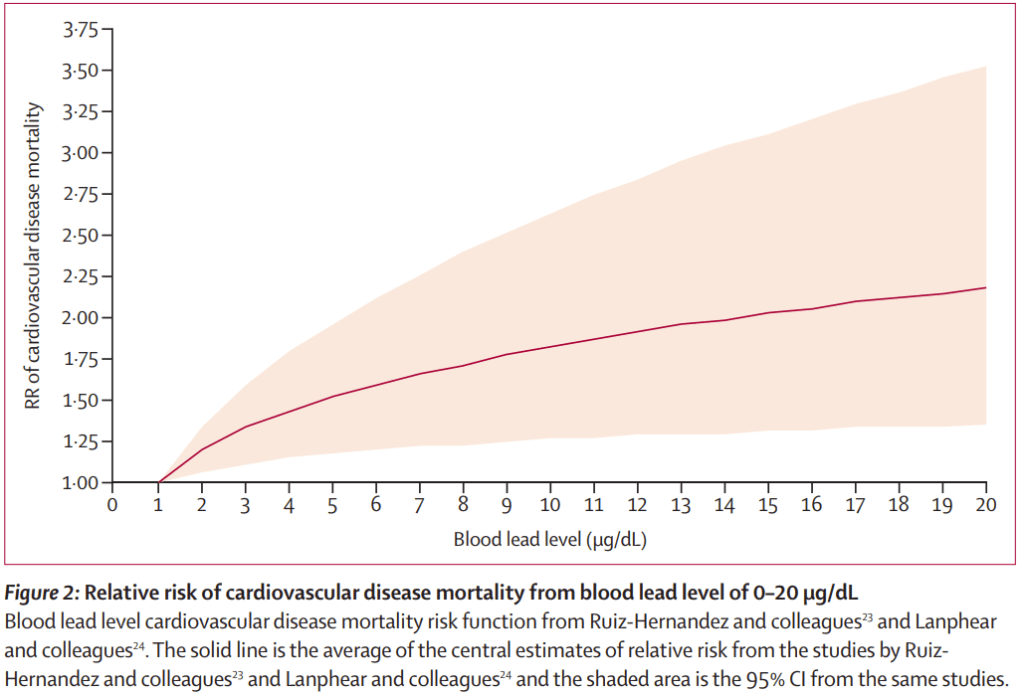
Using epidemiological data from the US CDC and adjusting the numbers to Australia’s population (about 28 million), some 30,000-40,000 Australians die every year from lead comorbidities, diseases where lead was a contributing factor. That’s including cardiovascular disease, kidney disease, neurological disorders and cancer. The death rate from lead is 10 times the death rate from asbestos exposure.
We absolutely can’t avoid lead exposure, and as things stand, neither can we avoid the health impacts caused by lead exposure. Lead accumulates in our bodies, either quickly or slowly depending on exposure, and as we age, the lead-caused medical conditions increase.
If we could remove lead from the environment, and from people, we would be saving a lot of lives and a lot of misery every year. Chelation offers a way of removing lead from people.
One of the biggest obstacles facing the widespread adoption of chelation treatment is that there is so much controversy about chelation that needs to be answered.
Nasty stuff aside, if you do an internet search (Google) with the phrase “is chelation dangerous”, you’ll find a lot of web sites have similar information, including the Harvard Medical School, namely:
Chelation can be dangerous, it can cause kidney and liver disease, and even death.
That’s pretty straightforward, but it lacks some critical supporting information beside the fact there are many variables in the medical use of chelating agents.
Most of the negative hype about chelation is due to the use the salts of ethylene diamine tetraacetic acid or EDTA so here is the history of the use of EDTA.
EDTA was originally synthesized in 1935 in Germany for use in the dye industry, but it’s preservative properties led to it being approved as a food additive by the US Food and Drug Administration in 1947, and it was also soon recognised as an effective way to remove heavy metals from the body.
Intravenous (IV) sodium EDTA administration was observed to have a useful side-effect, besides removing lead, because patients treated with EDTA also had relief from angina. A study by CN Clarke et al, published in 1956 (Am J Med Sci. 1956 Dec;232(6):654-66) indicated EDTA treatment resulted in a reduction of angina and abnormal ECG.
This is where things get a bit complicated because sodium EDTA gained another role, removing atheromas or atherosclerotic plaques. When we are talking about EDTA chelation, it could be EDTA being used to remove heavy metals from the body, or EDTA being used to remove calcification, not just for treating cardiovascular conditions but also hypercalcemia and conditions like tendon calcification. I think it’s crucial to make this point here because this distinction is often not considered in ill-informed medical circles.
The observation that chelation benefitted people with heart disease started a huge surge in treatment with EDTA where some patients did indeed exhibit kidney damage, later found to be due to too high cumulative doses. Protocols were soon established that avoided ill effects and EDTA was in fact found to have beneficial effects on kidney function.
Sodium EDTA was initially used for both removal of lead and treating calcification, but the removal of calcium that occurred, unless carefully monitored, could result in hypocalcemia and even death.
Calcium sodium EDTA removed lead as effectively as sodium EDTA and it wasn’t long before it was stipulated that calcium EDTA was to be used for removing heavy metals to avoid the risk of hypocalcemia.
Nevertheless, there were still deaths due to chelation with EDTA, but the ones reported were caused by the inadvertent administration of sodium EDTA instead of calcium disodium EDTA, sometimes due to a pharmacy error because the two salts had similar commercial names. Using calcium sodium EDTA (CaEDTA) results in a net increase in circulating calcium, so it can’t cause hypocalcemia and is safe for treating lead-poisoning, particularly in paediatric use.
The death of a 5-year old autistic boy being treated to remove heavy metals is often quoted as an example of the dangers of EDTA. He tragically died of hypocalcemia due to a pharmacy error, because sodium EDTA was supplied instead of calcium disodium EDTA because they had similar commercial names.
Two points have to be stressed here. CaEDTA administration would not have caused hypocalcemia, and if every drug that was mis-prescribed and caused death was banned, we would have a very restricted suite of pharmaceuticals available for patient treatment.
When IV administration of CaEDTA is considered as unacceptable or too dangerous, as for example with very young lead-poisoned children, it can be administered by intramuscular injection (IM). IM administration of CaEDTA is quite painful unless it is co-injected with procaine. Indian physicians have found that IM injection of magnesium EDTA is far less painful and equally effective for heavy metal removal and the dose avoids hypocalcemia.
CaEDTA can also be administered orally, but a frequent argument for using other chelators instead of EDTA salts is that EDTA is poorly absorbed in the gut. Early absorption studies did indeed indicate that the absorption of EDTA was relatively poor, about 5%. The reason for the poor absorption is due to the acid environment of the stomach which renders EDTA nearly insoluble. If taken with food , or in slow-release capsules, absorption can be as high as 15-20%.
It has also been argued that EDTA is relatively non-specific in terms of metals bound. This needs to be put in context.
There is a hierarchy of metals that will bind to EDTA, depending on the binding affinity between EDTA and the metal. Lead binds very strongly, followed by calcium, other heavy metals, magnesium and zinc. A stronger binding metal will displace a weaker one.
Depletion of essential metals is a valid concern, but it’s important to point out that the depletion is EDTA-dose dependent. If the dose of EDTA in the body is low enough, the depletion of essential metals is minimal. Trace metal supplementation is mandatory for IV chelation, may be necessary for IM chelation, but is generally unnecessary for oral chelation. Nevertheless, it would be prudent to include an occasional trace metal supplement regardless.
Now let’s briefly discuss other chelators that are alleged to be safer because of reduced trace metal depletion.
In general terms, there are 2 classes of chelators that bind lead, ones that contain sulphur, which has a great affinity for lead, and ionic chelators like EDTA. Absorption is generally much better in the sulphur-containing chelators but they do have a drawback as they are often partially metabolised in the body. This can result in unwanted side effects in some cases, for example with DMSA (dimercapto succinic acid) and skin reactions. But sulphur-based chelators do have a somewhat reduced affinity for essential minerals.
Despite the fact that other chelators such as DMSA and Penicillamine have different relative specificities for essential metals, the differences are unlikely to be significant at the relatively low doses used for oral treatment.
As always, there is an exception to the above “classes” that is in effect a combination of both classes of chelators, trimercapto triazine (TMT) which has an extremely high binding affinity for lead and isn’t metabolised in the body. TMT binds lead so tightly it is biologically unavailable, so it has been used to remove lead from cattle and to decontaminate soil. However, human trials have not yet been carried out to establish its safety for human use.
The development of custom circular polypeptide chelators that are much more specific for lead could have potentially avoided any essential metal depletion, but the significant extra cost of what is in effect a patented drug which still needs clinical trials means that practically speaking, CaEDTA with occasional mineral supplementation is much more cost-effective on a large scale.
I found that there was another EDTA salt in quite widespread use that had escaped my attention because it was being used for a totally different purpose. Ferric sodium EDTA, (FeEDTA), has been used in France as a paediatric iron supplement for nearly 50 years. It turns out that FeEDTA is the only iron supplement that provides bioavailable iron despite the presence of phytates that are found in many grains, and the polyphenols found in pulses.
The implications of this are that with a low meat or vegan diet , iron naturally present in vegetables is rendered unavailable. In the absence of bioavailable iron in the diet, the result, particularly in females, is iron deficiency.
An expert panel on flour fortification concluded in 2008 that FeEDTA was the only suitable form of iron to fortify whole grain (high extraction) flour in public health fortification programs. In 2009, this recommendation was endorsed by the World Health Organization. In January 2010, the Panel on Food Additives and Nutrient Sources added to Food (ANS) of EFSA concluded that ferric sodium EDTA as a source of iron at the proposed usage level in fortified food for the general population “would not be of safety concern”.
In the EU in 2010 was stipulated that “fortified food” can contain FeEDTA up to an amount that does not exceed 12 mg of EDTA calculated as EDTA‑H4. This corresponds with 2.3 mg Fe as FeEDTA which is sufficient to allow the fortified food product to be labelled as a “source of iron”.
Numerous investigations, including large field trials, have shown that FeEDTA is safe for human consumption at levels intended in food fortification . The limits imposed offer a 300-fold safety margin before the possibility of ill effects.
One of the challenges facing any large-scale lead detoxification is one faced by many drugs, and chelators are no exception. But CaEDTA and FeEDTA have already been tested in long term large-scale trials, as food additives. In Australia (and New Zealand) both salts are registered food additives, CaEDTA (#E385) and FeEDTA (#A570).
Iron deficiency exacerbates lead poisoning in young children, and is contributed to by lead poisoning. Iron deficiency also adds to the detrimental neurological effects of lead poisoning. If you treat one, you really need to treat the other, especially in children.
The real utility of FeEDTA was revealed in a large scale double blind study of children in Morocco. The results showed FeEDTA resolved iron deficiency AND also reduced blood lead levels over the 6 months of the trial. That result should perhaps have not been all that surprising since we know oral EDTA can remove lead, but both results at once is a bonus. Administration of FeEDTA to the children was as biscuits containing FeEDTA.
It should be quite clear that we have two food additives, one a preservative and the other an iron fortificant, that when added to food could potentially provide community immunity to lead exposure in the same way that folate added to food prevents spina bifida.
But using CaEDTA and/or FeEDTA in food isn’t quite that simple. We know that FeEDTA is endorsed by the WHO, but we can’t ignore the medical perception that chelators are nasty stuff, and of course the usual commercial and political considerations.
I was awarded my PhD working on iron metabolism in rats and humans, so iron metabolism is pretty familiar to me.
Most cereals, (oats, wheat, maize (corn), rice, sorghum, rye, and millet) contain phytates. Breakfast cereals are often “fortified with iron”, which ought to be good, but the form of iron is iron filings (finely ground food grade iron, also called reduced iron or ferrum reductum). Reduced iron is at best a poor source of bioavailable iron and if you add phytates or polyphenols, the amount of bioavailable iron is minimal.
Introducing FeEDTA to common foods seemed like an ideal way to start protecting the community from lead. But for one small problem.
I approached all major breakfast cereal manufacturers, pointing out the deficiencies of iron filings, and suggesting that they use FeEDTA, so their cereals would in fact be “a source of iron”.
Alas, when there is a choice of iron fortificants, the inevitable choice is for the least expensive ones. That means manufacturers continue to use reduced iron or iron sulphate and will not change unless legislation is enacted that specifices the iron must be bioavailable iron rather than just iron.
Until then, we can’t force manufacturers to make healthy food.
CaEDTA IS in fact being used as a preservative in foods like mayonnaise, salad dressings, sauces, margarine, spreads, and some canned foods like vegetables or seafood. That has to be seen as a Pyrrhic victory and an indication that maybe it isn’t quite such nasty stuff.
That’s probably a good place to close for now. I hope I’ve made you realise that the nasty stuff is in fact pretty good. If its use were more widespread, the threat of increasing bone lead levels and the accompanying cardiovascular disease, kidney disease, neurological disorders, gout and so on might be a lot less prevalent in our community.
If only.
In the interim, if you’re worried about the effects of lead on your loved ones, contact me and we can talk about what can be done. At this point in time there are a bunch of people taking oral CaEDTA to remove lead from their bodies, or FeEDTA in biscuits, and a small trial starting up in Broken Hill, run by mothers concerned about the effects of lead on their children.
Government agencies really aren’t interested in doing a lot beyond managing public opinion.
We CAN do something, there IS hope, but if you’re waiting for someone else to take the initiative, it’s a long wait.